Electrolytes and NonelectrolytesAll ionic compounds are electrolytes. Those that do not conduct an electric current are nonelectrolytes. Because they do not have ions most are molecular materials. There are strong electrolytes, and weak electrolytes depending on the degree of ionization.
|
The Solution ProcessThe solution process is called "solvation." The solvent takes the solute particles apart and surrounds, or solvates, them as a solution forms. The solvent molecules must come in contact with solute in order to dissolve. The types of solutions are saturated, unsaturated, and supersaturated. Saturated solutions hold the maximum amount of solute dissolved. Unsaturated solutions can dissolve more solute. Supersaturated solutions can hold more solute than it should.
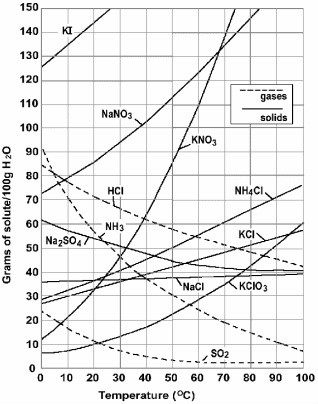
The graph below is a solubility graph. |
How many grams on KNO3 can dissolve in 100g of water at 50 degrees Celsius? 85g. You would find where KNO3 is, then look for the temperature at the bottom of the graph, after that you would go up the graph until the curve of KNO3 hits. It hits around 85g.
Definitions
The measure of the amount of solute dissolved in a solution is the concentration of a solution. A dilute solution contains a low concentration of solute. On the other hand, a concentrated solution contains a high concentration of solute. To compare solutions and express concentration we use molarity and percentages. We can make a dilution by using more solvent to a more concentrated solution.
Molarity of SolutionsMolarity (M) is the number of moles of solute dissolved per liter of solution.
moles of solute / liters of solution Example: 2.3 moles of sodium chloride, NaCl, in 0.45 liters of solution. 2.3mol/0.45L= 5.1M |
Concentrations of SolutionsWhat is the concentration of a solution that has 0.25 mol dissolved in a 125 mL of water?
1L/1000mL= 0.125L 0.25mol/0.125L= 2.0M |
Dilutions of SolutionsThe dilution reduces the moles of solute per unit volume.
M1 x V1 = M2 x V2 Example: If you dilute 175 mL of 1.6 M solution of LiCl to 1.0L, determnine the new concentration of the solution. M1= 1.6 M M2= ? (1.6M)(175)/1.0= 4.8M V1= 175L V2= 1.0L M1xv1/V2= M2 |
State University, North Carolina. "CUSTOMER SUPPORT." WebAssign. North Carolina Department of Public Instruction, n.d. Web. 30 May 2014."Chemical Cleaning." JV Industrial Companies Ltd.: Turnarounds. Ed. Joe Keon. JV Industrial Chemical, n.d. Web. 29 May 2014.